Making Washing Soda from Baking Soda
Can we make washing soda at home?
My seriously flawed pH test with unexpected results
(costs were updated 10-31-12)The last few months, I've been making my own laundry soap in my kitchen. I've used it long enough now to be very satisfied with the cleaning power and the fabric care of my family's clothing.
So far, I've been using baking soda as part of my laundry soap recipe. I noted that many of the recipes use washing soda and I wondered if that would not only be a cheaper alternative, but whether it would make an even more effective laundry soap.
Baking soda is technically sodium bicarbonate and washing soda is sodium carbonate. Washing soda is more alkaline than baking soda and therefore harsher. In fact washing soda is nearly caustic and should never come in contact with skin and never, ever ingested (eaten). However, it's that extra alkaline property that gives washing soda the ability to both soften water and clean better.
When sodium bicarbonate (baking soda) is heated, carbon dioxide and water are released and sodium carbonate (washing soda) remains.
When I did a search, multiple sites were indicating that it was possible to make washing soda from baking soda simply by heating baking soda in the oven at 375 to 400 degrees F for one or two hours. I wondered if this was real or just an urban myth that kept getting passed around. It took some digging to find out.
One website (MakingDoWithTheNotSoNew.com) had posted a discussion of making washing soda from baking soda. In the comments section, a reader named Jessica even commented that her chemist husband declared it impossible to make washing soda by heating baking soda. (Sorry, Jessica, he must have missed a few classes, keep reading ...)
I was further puzzled by an article on About.com, posted by Anne Marie Helmenstine, Ph.D. who basically said to heat baking soda in a 200 degree F oven for an hour and sodium carbonate would result. I respected the Ph.D., but was puzzled by the instructions to heat at 200 degrees for an hour, since my own experimenting and multiple posts indicated that the oven needed to be at least 300 degrees F for a minimum of one hour. So I was not sure if I could trust this article, either.
More searching and I found a photographer's site (Photo.net) discussion about how to make your own sodium carbonate for processing film. They were making theirs by heating the baking soda on the stovetop in a pan. (I haven't tried and don't recommend this method. They indicated you must cover the pan as the mixture will make little erupting volcanoes.)
At this point, I'm convinced that baking soda can, indeed, be heated to create washing soda. After all, these photographers were using it to develop their photos and needed a specific chemical (sodium carbonate) for the process.
Now I'm curious, mostly convinced, but still no solid research. So, I called the Arm and Hammer Company customer service line. I spoke with a very nice rep named Heather. At first she came back and said, no, it's not possible to make baking powder by heating baking soda. After correcting her, and also explaining the chemical terms of sodium bicarbonate to sodium carbonate, she put me on hold for a few more minutes. She came back with the reply, "That is not a recommended use and we have no comment." Hmmm....
I finally stumbled on a discussion in AbsoluteAstronomy.com regarding the process of creating sodium carbonate. One method was called the Solvay process.
At last! The Solvay process is a basic method used in factories and is apparently quite well accepted. This process starts with sodium choride and ends with sodium carbonate (plus a separate byproduct that was then converted back to ammonia for reuse). Here is the important line of the Solvay process:
"The sodium bicarbonate was then converted to sodium carbonate by heating it, releasing water and carbon dioxide:
2 NaHCO → Na2CO3 + H2O + CO2 "
Translated, this means that baking soda (NaHCO3) was heated to create washing soda (Na2CO3) plus water plus carbon dioxide. The water and carbon dioxide steam off during the heating pocess.
I was able to verify the Solvay process on multiple scientific sites. Unfortunately, I also found that the Solvay process heated the sodium bicarbonate to 300 degrees C, which is 572 degrees F. This would be a dangerous temperature to use at home.
So now I was really curious if the oven heating process of 375 degrees F really creates washing soda. Since washing soda is more alkaline than baking soda, I decided to test the powder that I had created by heating baking soda in my oven, to see if the pH was raised.
Baking soda has a pH of 8.4 and washing soda has a pH of 11.6, so if heating the baking soda does create washing soda, testing the pH should show a significant increase after heating.
To begin, I measured exactly one cup of baking soda and heated it in a glass baking dish in the oven at 375 degrees F. After almost three hours and occasional stirring, the powder was uniformly changed from fine-powder pure white to a slighly courser, cream-grey colored powder. I let this cool overnight.
As I started the experiment, I measured this new powder and found that it measured nearly 1-1/4 cups. This was puzzling, since I had started with 1 cup of baking soda and the heating was suppossed to release water and carbon dioxide, which should make it weigh less and I thought it would also make less volume. I left that question for another time.
For my experiment, I used pH tape, two glass jars, filtered water, and a tablespoon of baking soda and a tablespoon of the (hopefully) homemade washing soda.

Photo by Patti Tokar Canton © 2011
I had a roll of pH testing tape from Swanson Health products. I had just gotten it for free: on my last order they had sent the tape instead of the bottle of supplements that I had ordered. Seems Swanson now has robots roaming the warehouses and packing the orders (cool, huh?) and the robot couldn't tell the differenct between a box of tape and a bottle of capsules. At any rate, Swanson was polite and efficient when I called and they immediately sent a shipment of the supplement and graciously told me to keep the pH tape. So it was this coincidence of getting the free pH tape that started this entire experiment.
To start the experiment, I measured filtered water into a clean glass jar. I then tested the pH of the water as a control.

Photo by Patti Tokar Canton © 2011
It tested between 6.4 and 6.6, showing a slightly acidic water. Neutral is 7.0.
Next, I added a tablespoon of Arm & Hammer Baking soda to the water.

Photo by Patti Tokar Canton © 2011
Note that the baking soda is very fine and a little lumpy.
I then shook and swirled this solution until the baking soda was dissolved. Then I tested the solution with the pH tape. The tape turned a deep purple. This is where I discovered the fatal error of the experiment. When I tried to read it, I found that my chart only went to 8.0. I was further dismayed to see that my tape was only rated to go to 9.5 pH. Since I was looking to test 8.4 (baking soda) and 11.6 (washing soda) I now realized I did not have the right testing tape and would not be able to really know how much the pH of the heated powder had changed.
Not easily dismayed, I continued on.
Now, I got another glass jar, added the same amount of filtered water and tested the pH of the plain water. I got about the same reading, this seemed more 6.6, but very similar to the first jar of water.
Here are the three tapes so far:

Photo by Patti Tokar Canton © 2011
From left to right: First filtered water, After adding baking soda, Second filtered water
Next, I added a tablespoon of the (hopefully) washing soda.

Photo by Patti Tokar Canton © 2011
Note how the powder is grainier, yet uniform.
I shook and swirled this mixture, then tested it. The test strip instantly turned a deep, dark purple - the reaction was much faster than the baking soda test.
I compared the test strip to the chart and to the baking soda test strip. It was darker, but obviously I had maxed out the test strip.

Photo by Patti Tokar Canton © 2011
The photo does not really show the difference. Now I'm discouraged. I can tell that the pH of the (hopefully) washing soda is more alkaline than the baking soda, but I don't know how much, and certainly not enough evidence to determine that it is close to the 11.6 that I need to show that it had reached the pH of washing soda.
Still, I called in my husband, Chris, to look at the strips. His background is in graphic design and color matching and he currently runs a painting business where he uses his color skills regularly. I asked him to look a the strips and give me his estimate of the comparison of color shades of the strips.

Photo by Patti Tokar Canton © 2011
He determined that the fourth strip was at least 4 progressive shades darker than the second strip, which would mean that the homemade washing soda was more alkaline than the baking soda.
So while I have no measure (until I get new test strips) to tell if I got the pH to 11.6, I can readily see that heating the baking soda did change the alkaline level.
Then I made the discovery that made the whole experiment worthwhile. Note the photo below of the two tested jars:
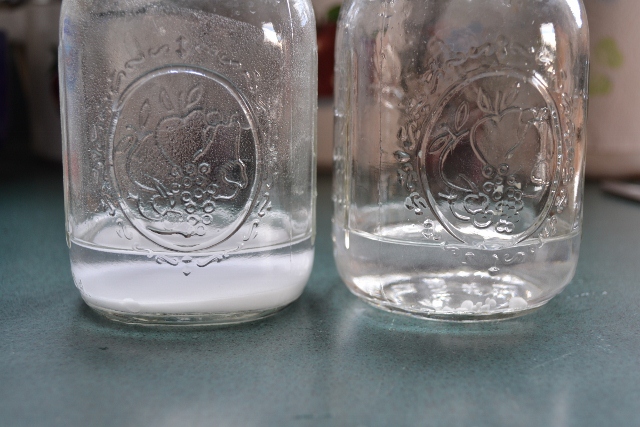
Photo by Patti Tokar Canton © 2011
The jar on the left is the baking soda and water mixture. The jar on the right is the homemade washing soda and water mixture. Note the entire bottom of the baking soda jar is lined with undissolved baking soda! Now look at the homemade washing soda jar. The water is crystal clear!
To test this, I shook and swirled the baking soda jar for another minute or two.
Here is a photo from an overhead view:

Photo by Patti Tokar Canton © 2011
Again, the jar on the left is the baking soda, and still has a thick layer of white on the bottom. The jar on the right is the homemade washing soda and if you look close toward the bottom of the jar in the photo, you will see that the only reside is a sprinkle of little white "rocks", which I theorize is pieces of baking soda that did not get heated enough to transform the baking soda.
This crystal clear liquid would certainly be much better at rinsing away from clothing in the washer. Remember that I mixed the baking soda jar twice and the homemade washing soda only once. Heating dramatically changed the dissolving properties.
Conclusion:
1. Heating baking soda does increase the alkaline properties, although I was not able to determine how much. I can unscientifically say that the homemade washing soda appeared to be substantially more alkaline, based simply on the deep and instantaneous change of the pH tape color.
2. Heating baking soda definitely changes the dissolving properties of the resulting powder. The homemade washing soda dissolved in water with no effort. The original baking soda did not fully dissolve after a second mixing.
Comparing Costs:
Assuming that I did create genuine washing soda (sodium carbonate), was it worth the cost savings?
The baking soda was purchased at Sam's Club. The current price (10-30-11) is $6.16 for a 13.5 lb bag. There are 7.78 ounces of baking soda in one cup. That makes the cost of one cup of baking soda equal $.2218.
Heating in the oven at 375 F for three hours adds a cost of $1.01 (assuming a cost of $.34 per hour of heating time)
Therefore the cost of the homemade washing soda in my experiment was $1.23 for about 1-1/4 cups. (Remember the volume increased when heating) One cup would be $.98.
Now, to compare this to washing soda purchased at retail, if you can find it. A 55 oz box of Arm & Hammer costs an estimated $2.99 (I haven't found it locally yet) The review section on Amazon.com for this product plus a listing on buy.com show this to be a reasonable estimate. This makes the price about $.054 per ounce. A cup of washing soda is approximately 8.4 ounces, making the total retail cost $.454.
Summary:
Cost of making one cup of homemade washing soda, without the cost of the oven heat: $.22
Cost including oven heat: $.98
Cost of one cup of washing soda purchased at retail: $.45
In my experiment, the cost of the homemade washing soda was more than twice as expensive as purchased washing soda.
Possible Changes to Change the Cost:
I could possibly reduce the cost by:
1) Making a large batch - at least five cups of baking soda at a time. In this case, I'd want to do some research regarding the effects of releasing this much carbon dioxide in the oven. Would it extinguish the gas flame? That could be dangerous.
2) Heating the baking soda in small quantities only when I am using the oven for something else, therefore getting "free" heat.
3) Experimenting with spreading the baking soda in a thinner layer in the pan, therefore possibly reducing the time it needs to heat in the oven.
4) Experimenting with the stove-top heating method discussed on the photography site. (I still don't recommend it until I can test the safety)
5) Buying the baking soda in much larger quantities: non-food grade sodium bicarbonate is available in farm supply stores. Mysuburbanhomestead.com reports buying a 50 pound bag for $13.99. That would bring the cost of the baking soda down to $.14 a cup.
Note: Alternatively, I could search a hardware or pool supply store for bags of sodium carbonate (washing soda) also called ash soda. A 100 pound bag is available at cqconcepts.com for $62.96 ($.33/cup) but I don't know what the shipping cost is. A 25 pound bag is available on eBay for $20.99 but the shipping adds $14.95 for a total of $.76 a cup. Not much savings here!
So, if you really want washing soda, it may make more sense to look in your local stores for the best cost. If you want to make your own, then try to heat it when you are already using the oven.* You will probably not have to heat it as long as I did if you use a larger pan and spread it thin. (In my experiment, I used a rather small baking dish and the baking soda was about 1/2" thick in the pan.) Do not use an aluminum pan, since sodium carbonate reacts with aluminum. Use a glass baking dish for best results. Experiment yourself to find the lowest heat and the shortest heating time for converting.
Remember to be curious and have fun saving money. That's the Fat Dollar way.
Article by Patti Tokar Canton.
All Rights Reserved. Do not duplicate without permission.
*Important Note: Please heat baking soda in your oven at your own risk! I don't have any information on the safety factors of this process and therefore can't make any recommendations on how safe the process is. Neither I nor The Fat Dollar will take responsibility for any results you may or may not have. Heating baking soda releases carbon dioxide gas and water vapor. The process creates a potentially caustic powder that should not come in contact with skin. It should also not come in contact with aluminum pans since sodium carbonate will corrode aluminum.
Resources:
MySuburbanHomestead.com - http://www.mysuburbanhomestead.com/find-cheapest-source-baking-soda/
Amazon.com - Arm & Hammer Super Washing Soda (read the reviews) - http://www.amazon.com/gp/product/B0029XNTEU/sr=8-1/qid=1319939534/ref=olp_product_details?ie=UTF8&me=&qid=1319939534&sr=8-1&seller=
Buy.com - http://www.buy.com/retail/product.asp?sku=215956159&listingid=86967656
Wikipedia - Sodium bicarbonate http://en.wikipedia.org/wiki/Sodium_bicarbonate
The Engineering ToolBox - pH values - http://www.engineeringtoolbox.com/bases-ph-d_402.html
City Collegiate - Chemistry of Sodium Carbonate - http://www.citycollegiate.com/sblock2.htm
PennilessParenting.com - http://www.pennilessparenting.com/2011/01/homemade-washing-soda.html
MakingDoWithTheNotSoNew.com - http://makingdowiththenotsonew.blogspot.com/2009/02/homemade-laundry-soap-with-baking-soda.html
About.com Chemistry - http://chemistry.about.com/od/makechemicalsyourself/a/Sodium-Carbonate-From-Baking-Soda.htm
DiaperPin.com - http://www.diaperpin.com/clothdiapers/article_bakingsoda.asp
Wikipedia - Solvay Process - http://en.wikipedia.org/wiki/Solvay_process
Charles Sturt University - Chemistry - http://hsc.csu.edu.au/chemistry/options/industrial/2765/Ch956.htm
Ausetute.com.au - http://www.ausetute.com.au/solvay.html
eNotes.com How Products Are Made - http://www.enotes.com/how-products-encyclopedia/baking-soda
Article by Patti Tokar Canton
© 2011 All Rights Reserved. Do Not Duplicate Without PermissionRECOMMENDED BOOK

Your Money or Your Life by Joe Dominguez and Vickie Robin
9 Steps to Transforming Your Relationship With Money and Achieving Financial Independence.
Are you working to make a living or working to make a life? Change the way you treat money and think about money. Revolutionary, inspiring and empowering nine-step program for transforming your relationship with money and achieving financial independence. This book is a classic and has changed many lives and Amazon readers give it a 4+ star rating (out of 5)!
 THE COMPLETE TIGHTWAD GAZETTE by Amy Dacyczyn
THE COMPLETE TIGHTWAD GAZETTE by Amy Dacyczyn
....This is one of my favorite books.
Although a little quirky sometimes, and sometimes a little extreme, this book has many great ideas for spending less on low priority stuff so you'll have more money to spend on high priority stuff (like college, or a house!) The author, Amy Dacyczyn lived her dream of having a large farm house and many happy kids all while working from home and learning how to be creative and frugal. Amy's background in graphic design makes her approach to saving money simply delightful. The best part for me was the wonderful tone of family love that weaves throughout the book. Very creative and fun book and VERY THICK! You will definitely get your money's worth in simple living tips and money saving tips with this book.

THE COMPLETE IDIOTS GUIDE TO BEING A CHEAPSKATE by Mark W Miller. Help with saving money on travel, gifts, home items and more in the traditional "complete idiot's guide" format.
Books are offered in association with AmazonThe Fat Dollar Ideas
Vitamins and Fruit Juice - save $63.87 a year and 20,440 calories
Recipes for Stale Marshmallows
How to Re-Crisp Stale Ready-to-Eat Cereal
Save Money on Food with Inexpensive Clothespins
In the Home - tips and ideas to save money in your home
Make Your Own Shampoo and Rinse - Herbal or Plain
Save Money on Anti-Aging and Skin Care Creams
Stop Comparing and Just Enjoy Your Life
Self-Employment - The Fat Dollar will give you support and ideas to increase your income and still let you live a joyful life
Manage Your Money - Tips for managing your Fat Dollars
How to Stop Impulsive Spending
Save Money on Gasoline - Increase Your Mileage Up to 26%
Let Uncle Sam Help Finance Your Retirement Plan
Bartering Online or Offline - To fatten up your dollars
Fat Dollar recommends Simply Free Stuff site - Free samples and products
Books to help you save money and regain control of your Fat Dollars
Save Grocery Money Books - free bonus cookbook with purchase

